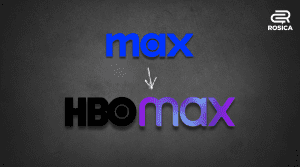5 ROI-driven PR Tips for Animal Health Companies and Veterinary Practices
Animal health companies and veterinary practices operate in a uniquely compassionate, complex, and challenging environment. [...]
Instagram’s School Partnership Program
In early 2025, Instagram introduced a new initiative aimed at combating cyberbullying and improving digital [...]
Top Marketing Strategies for Education Institutions and Ed Tech Companies
The education and ed-tech sectors are facing growing pressure to differentiate in an increasingly competitive [...]
Meta Brings Back “OG Facebook” – What Will this Mean for Education, Nonprofit, Healthcare, and Other Mission-Driven Organizations?
Meta recently took a nostalgic turn, reviving the essence of early Facebook by introducing a [...]
Content Marketing Best Practices for Animal Health Companies
The animal health industry is evolving rapidly, fueled by increased pet ownership, innovations in veterinary [...]
Advances in PR and Thought Leadership Measurement
In today’s digital-first, data-driven environment, the role of PR is evolving fast. For decades, organizations [...]
Four Media Training Imperatives in Today’s PR World
In a media landscape that’s faster, more fragmented, and more skeptical than ever, spokespeople and [...]
HBO Max Rebrands Again: Crisis Comms. and Branding Expert Chris Rosica Tapped by Business Insider for Commentary
In a move that’s drawing attention across the media world, Warner Bros. Discovery is rebranding [...]
Six Crisis Communication Tips for Veterinary Practices and Hospitals
In today’s high-pressure animal health and pet care environment, veterinary practices and hospitals face a [...]
Positioning & Messaging Your Brand in Today’s Digital World
In today’s hyper-saturated digital landscape, brands—both nonprofit and for-profit—are no longer just competing on product [...]
LinkedIn’s “Connection-only” Feed: A Game Changer for Professional Networking?
LinkedIn has long served as the digital meeting place for professionals, connecting individuals and organizations [...]
The Pros & Cons of Rebranding a Well-Known Name
Rebranding is no small undertaking—especially for nonprofit organizations with decades of name recognition, donor trust, [...]